Basics of Static Electricity
by
Ron Kurtus (revised
Static
electricity is the situation where electrical charges build up on the surface
of a material. It is called “static” because there is no current flowing as in
AC or DC electricity. Static electricity is usually caused when materials are
rubbed together. The result is that objects may be attracted to each other or
may even cause a spark to jump from one object to the other. Common examples of
static electricity in action are static cling, flyaway hair and the sparks that
can occur when you touch something.
Cause of static
electricity
Static
electricity is usually caused when certain materials are rubbed against each
other, like wool on plastic or the soles of your shoes on the carpet. The process
causes electrons to be pulled from the surface of one material and relocated
on the surface of the other material. The
material that loses electrons ends up with an excess of positive (+) charges. The material that gains electrons ends up an excess of negative (-)
charges on its surface. Electrons pulled from
orbit The
gain or loss of electrons can be explained by recalling that atoms consist of a
nucleus of neutrons and positively charged protons, surrounded by negatively
charged electrons. Normally, there is the same number of electrons as protons
in each atom. But
if some object pulls away electrons from their orbit or shell around the nucleus, that causes the atom to have a positive charge
because it has more protons than electrons. Likewise, the other material will
have extra electrons in its shell, giving the atoms a negative charge. Charges on surface Note
that the charged atoms are on the surface of the material. Static electricity
is different than regular electricity that flows through metal wires. Most of
the time the materials involved in static electricity are
nonconductors of electricity. If
electrical charges build up on the outside of a metal, most of them will
dissipate into the metal, similar to an electrical current. Prefers dry air When
the air is humid, water molecules can collect on the surface of various
materials. This can prevent the buildup of electrical charges. The reason has
to do with the shape of the water molecule and its own electrical forces. Thus,
static electricity is formed much better when the air is dry or the humidity is
low. Force field causes
attraction An
object that has static electricity charges built up on its surface has an
electrical force field coming from the surface. This field will mildly attract
neutral objects or those with no charge. The field will strongly attract an
object that has an opposite charge on its surface. From this we get the
expression: "Opposites attract." If
two objects have the same charge, the electrical force field will cause those
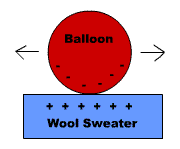
objects to push away from each other or repel. Attraction Rub
a balloon on a wool sweater. The balloon collects negative electrical charges
on its surface and the wool collects positive charges. You can then stick the
balloon to the wall, which does not have an excess of either charge. The
balloon will also stick to the wool, although the charges may jump back to the
original material in a short time. You
can also run a comb through your hair to charge the comb with static
electricity. The comb can then be used to attract neutral pieces of tissue. Picking up tissue with a comb Repulsion Comb
your hair on a dry day or after using a hair drier. The plastic comb collects
negative charges from the hair, causing the hair to have an excess of positive charges.
Since like charges repel, the hair strand will tend to push away from each
other, causing the "flyaway hair" effect. Why sparks fly When
two objects that have opposite charges get near each other, the electrical
field pulls them together. What
actually happens is that the negatively charged (-) electrons are attracted to
the atoms in the other material that have an excess positive (+) charge. Things
are much more stable if all the atoms have an equal number of (+) and (-)
charges. Strong forces hold
electrons The
reason the electrons can't leave their present material is because of strong
molecular forces that keep them where they are. If there are enough positive
(+) charges attracting them, and the distance is not too great, some of the
electrons will break loose and fly across the gap to the (+) side. Once it starts Once
a few electrons start to move across the gap, they heat up the air, such that
more and more will jump across the gap. This heats the air even more. It all
happens very fast, and the air gets so hot that it glows for a short time. That
is a spark. The
same thing happens with lightning, except on a much larger scale, with higher
voltages and current. Ben Franklin Ben
Franklin proved that lightning was static electricity by flying a kite in a
storm and detecting static electricity by seeing the hairs on the kite string
stand on end and creating a spark with a metal key. This dangerous experiment
showed that static electricity was being formed in the clouds by the rain. In conclusion Rubbing
certain materials together can cause the buildup of electrical charges on the
surfaces. Opposite charges attract and same charges repel. Either charge will
be attracted to something of neutral charge.